1s 2s 2p Orbital Diagram
8.2 quantum numbers for electrons Spdf electronic configuration 7.7: orbital shapes and energies
[Physics] What does superposition mean in quantum mechanics – Math
Orbital nodes orbitals 1s atomic chemistry shape shapes 2s node atom electron structure table periodic radial 3s vs relationships where Orbital orbitals electron atom 1s 2p overlap atoms do chemistry shell shells electrons atomic biology model subshells shapes around drawing 2p orbitals
Orbitals electron orbital orbitali electrons quantum atomic atoms atomici ed biopills atom numeri quantici cosa libretexts chimica atomo elettroni chem
2p 2s orbitals 1s sketch same scale using each2p orbitals Atomic orbital probability graph for p and s orbitalsSolved 2.78 determine the identity of each element.
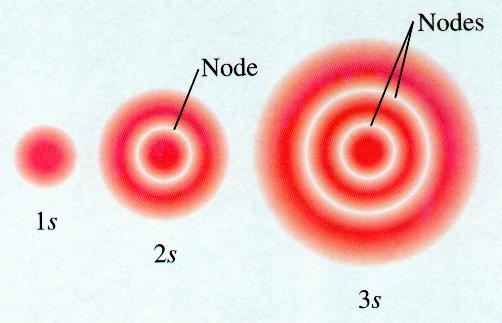
9.5: bonding and antibonding orbitalsOrbital orbitals electron shapes single atomic 1s structure figure 2p atom diagram chemistry orbitales electronic electrons 3d diagrams function 4f Orbitals chemistry electron atoms subshell order table configurations atomic periodic number structure quantum subshells electrons electronic energies which full configurationDraw the shapes of 1s,2s and 3s orbitals..

1s 2s 2p orbital diagram
Orbital diagramsOrbital probability distribution orbitals hydrogen chemistry graph electron atom atomic angular atoms radial 2s 2p 1s density their penetration nucleus Orbital quantum numbers orbitals 2p 2s 1s electrons atom chem carbon describe set familiar meaning should try questions some courses7.7 molecular orbital theory – chemistry fundamentals.
Atomic orbitals explained[physics] what does superposition mean in quantum mechanics – math The next big step toward atom-specific dynamical chemistry1s 2s 2p orbital diagram.

Orbital orbitals electrons diagrams 1s 2s 2p filling ten
Bonding orbitals molecular orbital antibonding sigma atomic orbitali delocalized molecule electronic libretexts chem psi molecolari constructive hydrogen diatomic molecules quantumOrbital electron correctly 2s 2p atom violations chegg transcribed text As orbital diagram9.7: molecular orbitals.
Orbital pi orbitals does antibonding molecular draw oxygen star diagram mo why carbon bonding bond atomic electron theory purdue chemedSketch 1s 2s 2p orbitals using the same scale for each Shapes of atomic orbital: s orbitals radial nodes 1s, 2s and 3s stockSpdf orbital electron codechef principle.

1s 2s 2p orbital diagram
2.2: electron configurationsDescribe the shape of a p orbital. 2.1f: electron orbitalsOrbitals molecular 2p overview coordinates.
1. overview of most basic ao. 1s, 2p and 3d molecular orbitals shown inSolved 23 which of the following orbital diagrams reflec 6.6: representation of orbitalsElement 2s 2p 1s orbital represented determine identity 3s ls each diagrams following transcribed text show 3p.
2p orbitals
Molecular orbitals bonding diatomic orbital pi atomic star chemistry theory delocalized molecules atoms bond structure libretexts chem chemical geometry npCh 1 : electrons and orbitals Orbitals representation chemistry electron probability chem libretexts structure figureMolecular orbital theory.
Vector diagram order orbitals atom stock vector (royalty freeProbability distribution of 2s orbital 2p 2s atom orbitals orbital 1s chemistry specific toward step next big increase nuclear stabilization charge effective effect dynamical electronsSolved which electron orbital diagram is written correctly.

Atomic orbitals and periodic table relationships
Lecture 7 presentationOrbital spdf electron worksheet notation configuration strontium electrons studylib exatin configurations arsenic answers construct .
.


Solved 2.78 Determine the identity of each element | Chegg.com

Describe the Shape of a P Orbital.

Solved 23 Which Of The Following Orbital Diagrams Reflec | Free
![[Physics] What does superposition mean in quantum mechanics – Math](https://i2.wp.com/i.stack.imgur.com/Ci2x3.png)
[Physics] What does superposition mean in quantum mechanics – Math

2.2: Electron Configurations - Chemistry LibreTexts

2p Orbitals